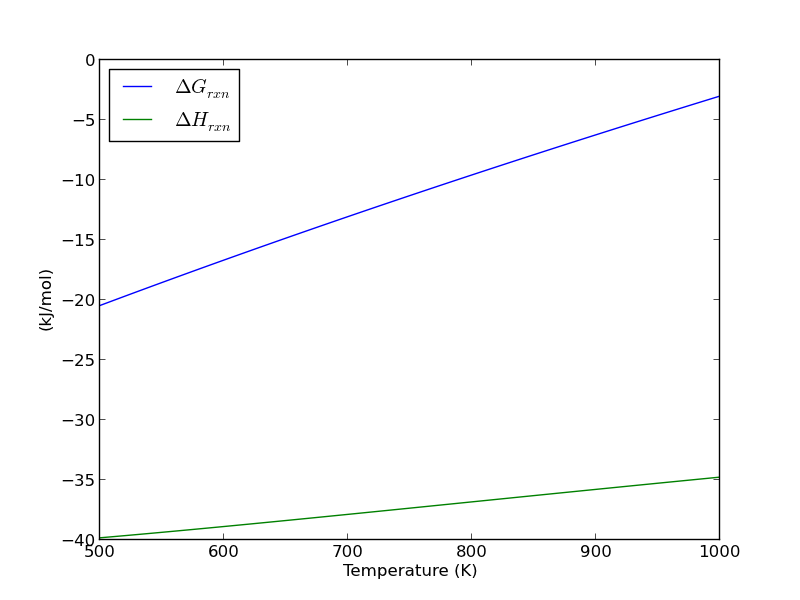
A novel formulation representation of the equilibrium constant for water gas shift reaction - ScienceDirect

Syngas Production via Reverse Water‐Gas Shift Reaction over a Ni‐Al2O3 Catalyst: Catalyst Stability, Reaction Kinetics, and Modeling - Wolf - 2016 - Chemical Engineering & Technology - Wiley Online Library
CO2 conversion by reverse water gas shift catalysis: Comparison of catalysts, mechanisms and their consequences for CO2 conversi

Role of water-gas-shift reaction in Fischer–Tropsch synthesis on iron catalysts: A review - ScienceDirect

Thermodynamic Analyses of a Moderate-Temperature Process of Carbon Dioxide Hydrogenation to Methanol via Reverse Water–Gas Shift with In Situ Water Removal | Industrial & Engineering Chemistry Research
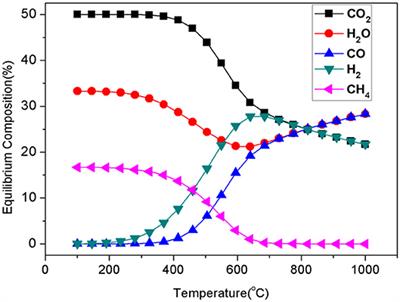
Frontiers | Recent Advances in Supported Metal Catalysts and Oxide Catalysts for the Reverse Water-Gas Shift Reaction
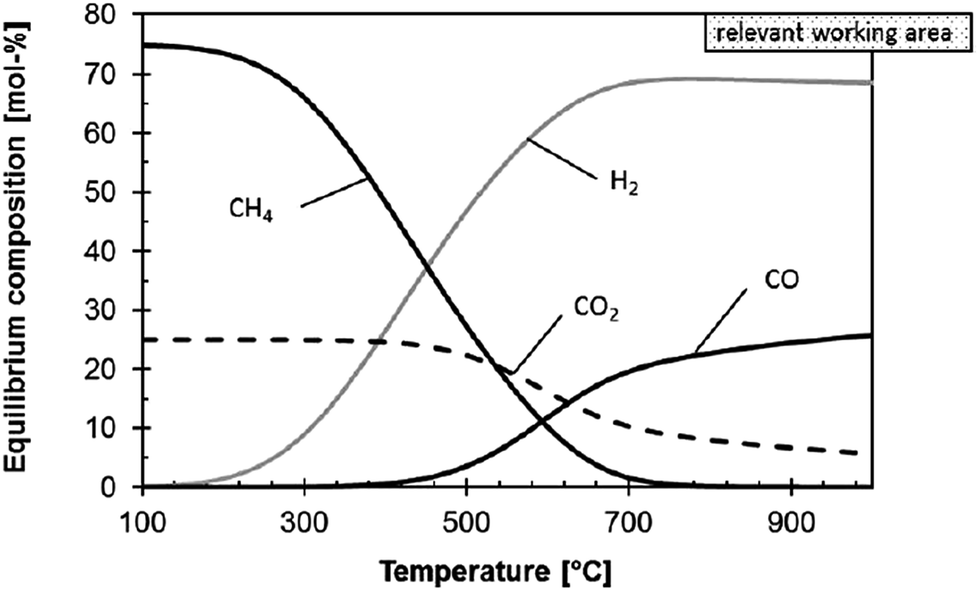
CO 2 conversion by reverse water gas shift catalysis: comparison of catalysts, mechanisms and their consequences for CO 2 conversion to liquid fuels - RSC Advances (RSC Publishing) DOI:10.1039/C6RA05414E

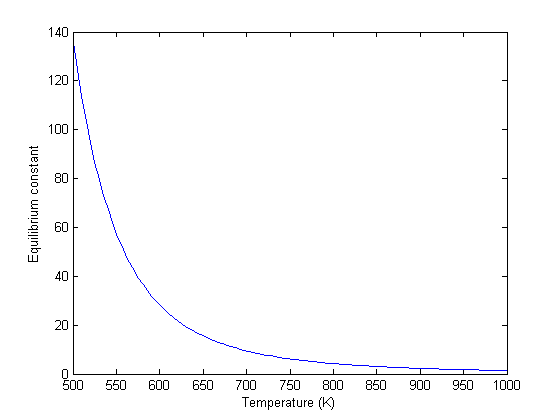





![PDF] A Review of the Water Gas Shift Reaction Kinetics | Semantic Scholar PDF] A Review of the Water Gas Shift Reaction Kinetics | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/dfcf4e0a08c25d179b68a5e0541881bfd7f07051/20-Table5-1.png)