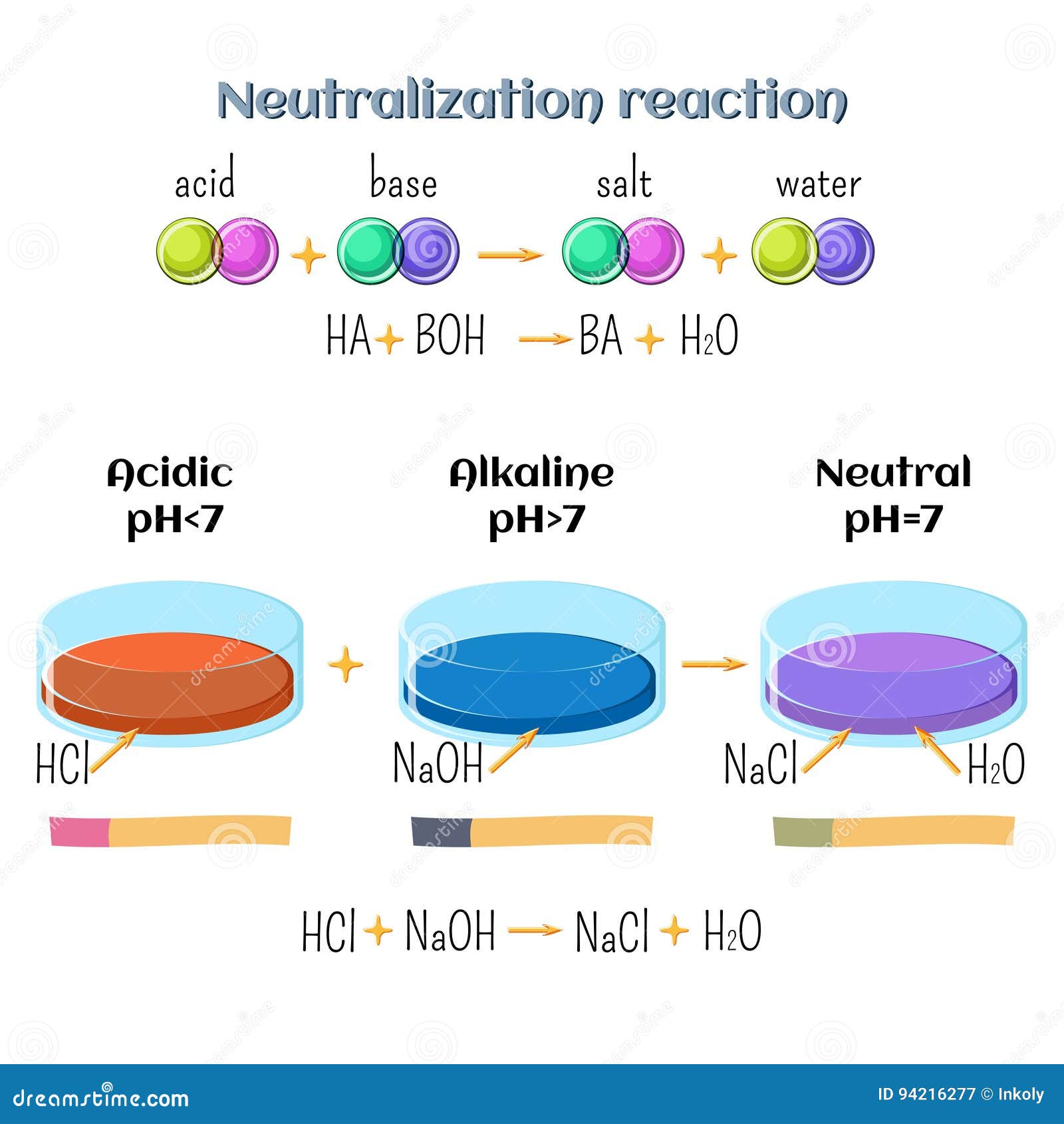
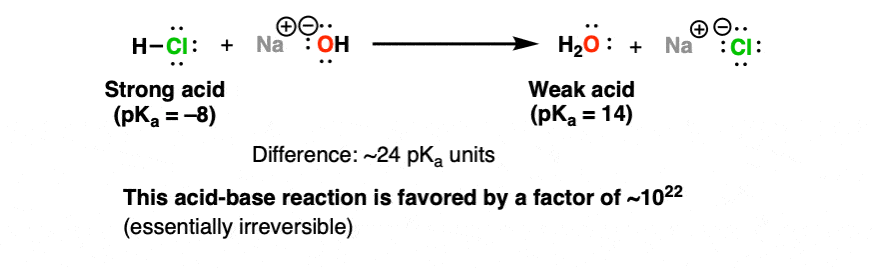
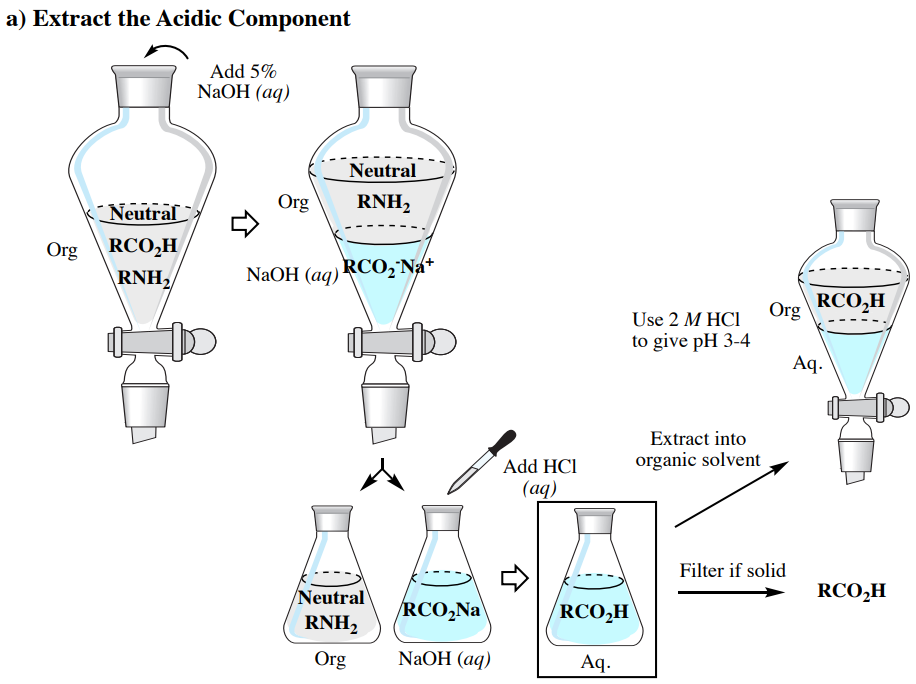
Réaction Acide-base. Réaction Chimique Neutralisant Les Propriétés Acides Et Basiques, Produisant Un Sel Et De L'eau. Utilisé Pour Déterminer Le Ph. Bronsted - Théorie De Lowry. Molécules De Hcl, Naoh, H2o Et

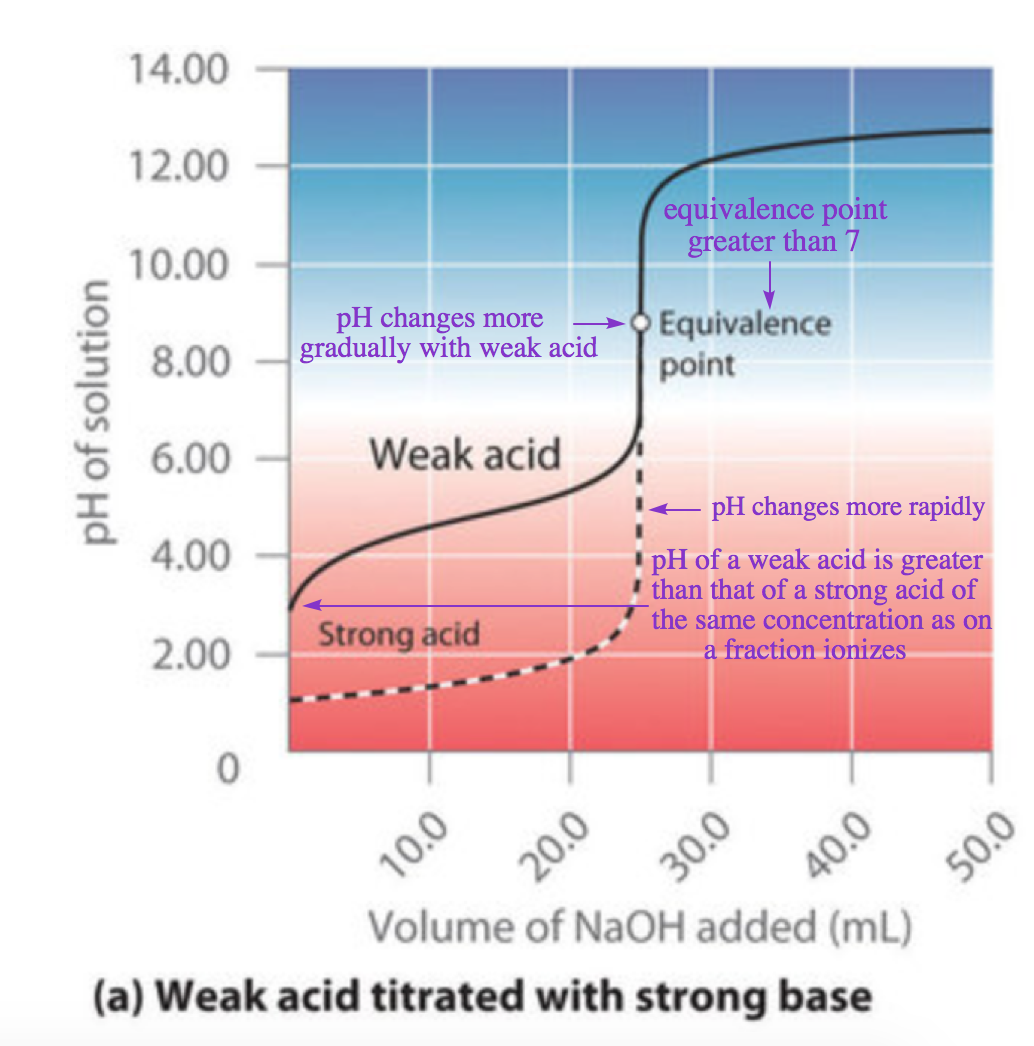
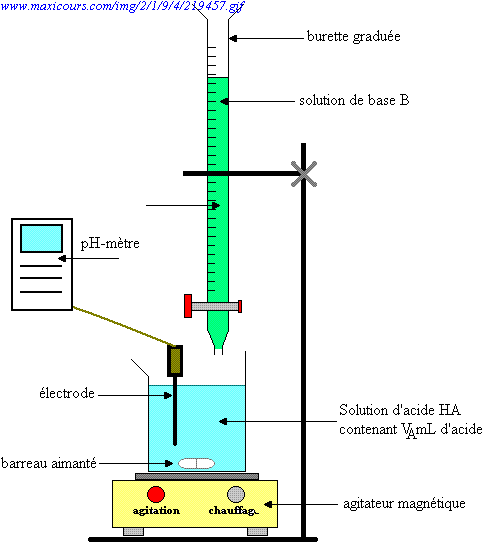
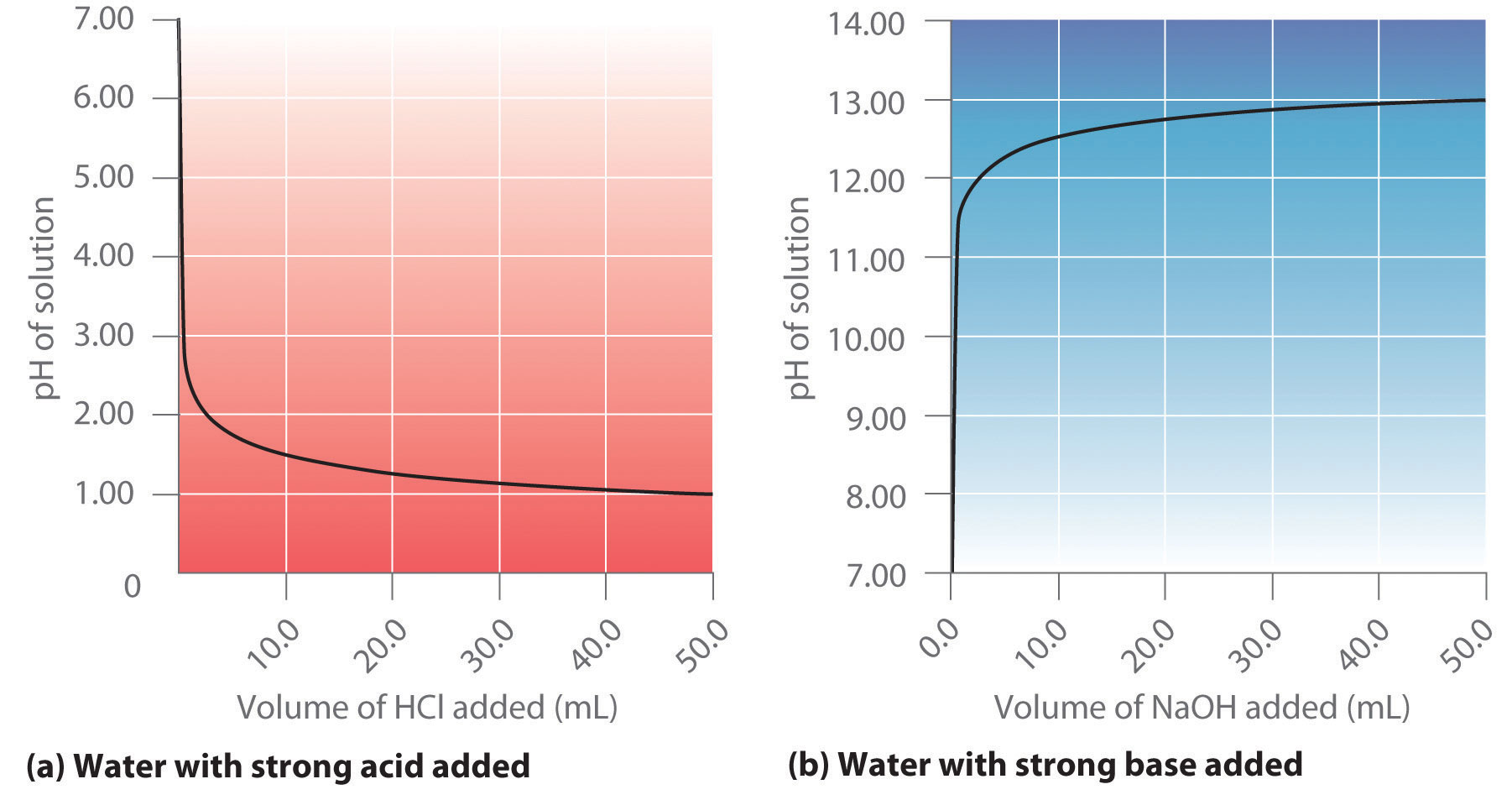
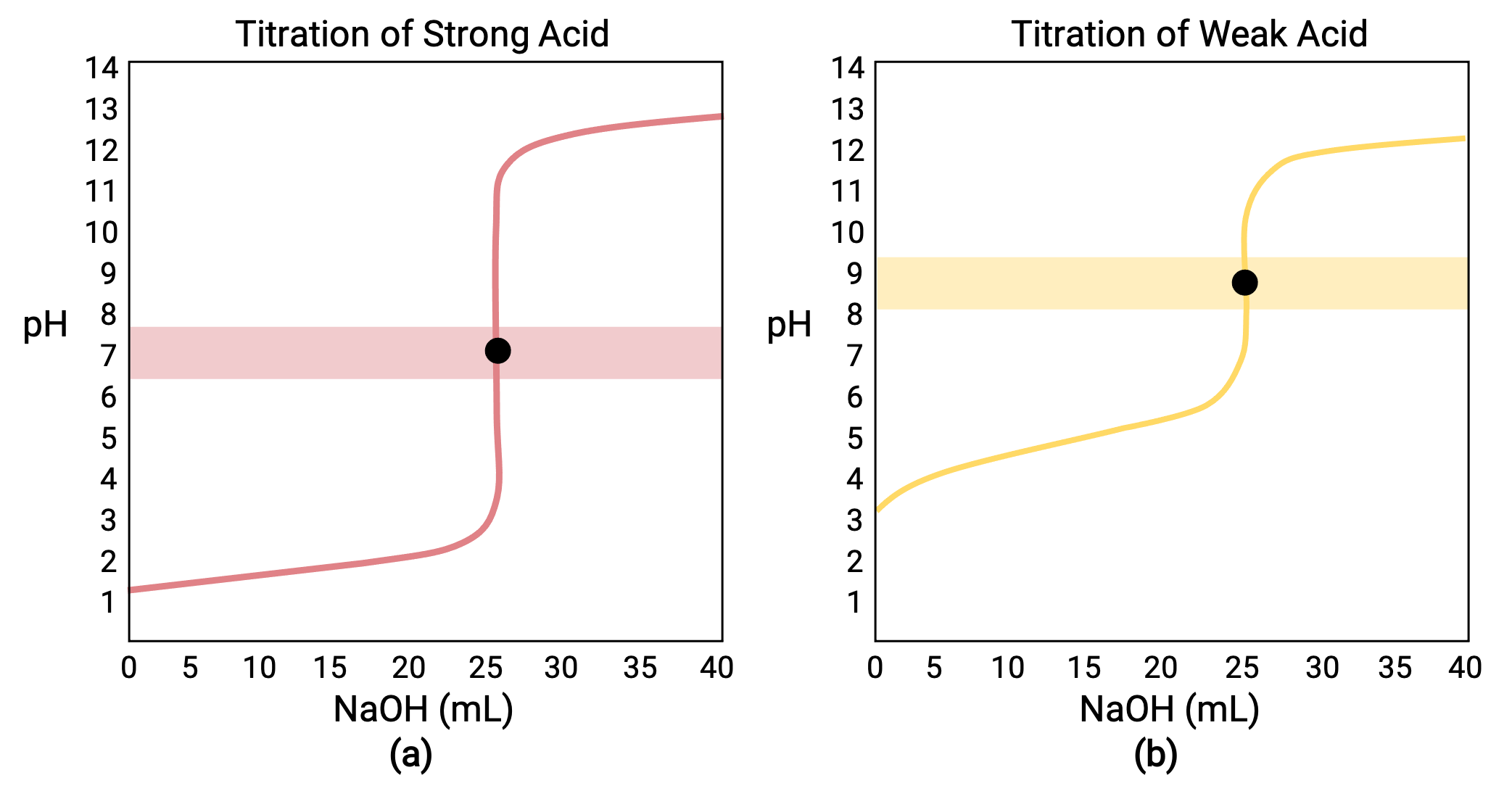
SOLVED: Question 4 (1 point) You will determine the concentration of an unknown diprotic acid using your standardized base: The chemical equation for this reaction can be expressed as: Vous determinez la

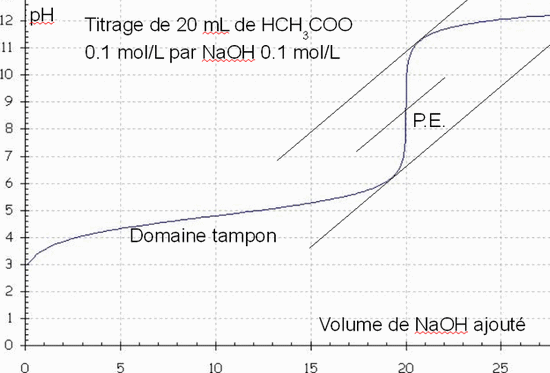
SOLVED: The balanced equation for the reaction between acetic acid and sodium hydroxide is shown below: HC2H3O2(aq) + NaOH(aq) à H2O(l) + Na C2H3O2(aq) Suppose that you perform a titration experiment to