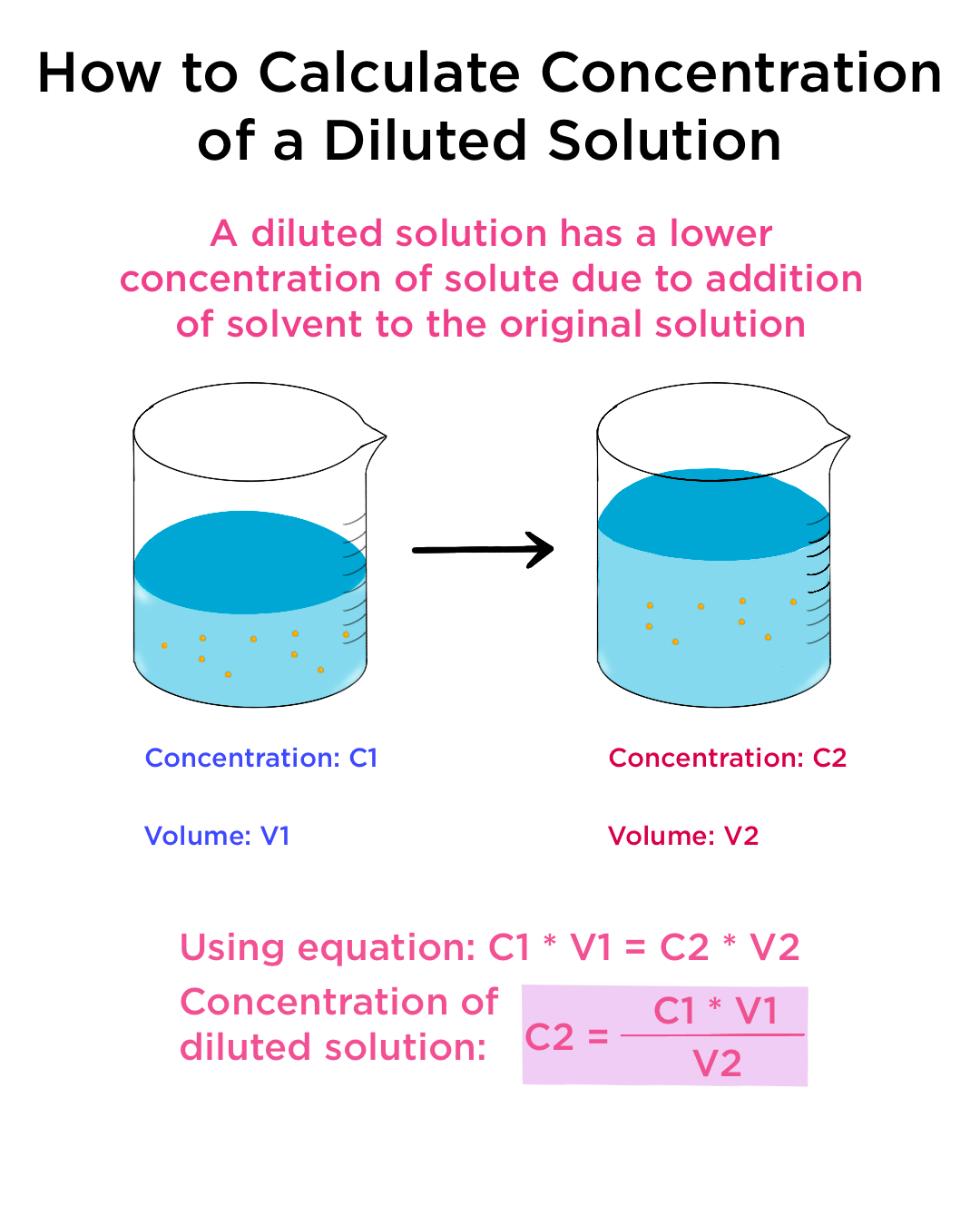
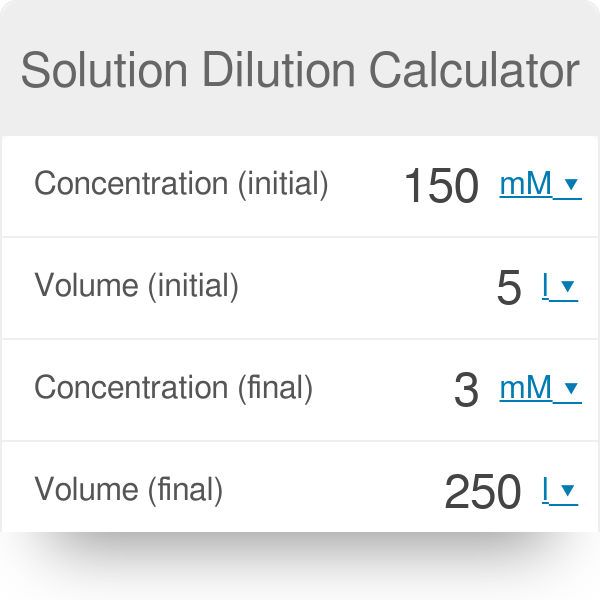
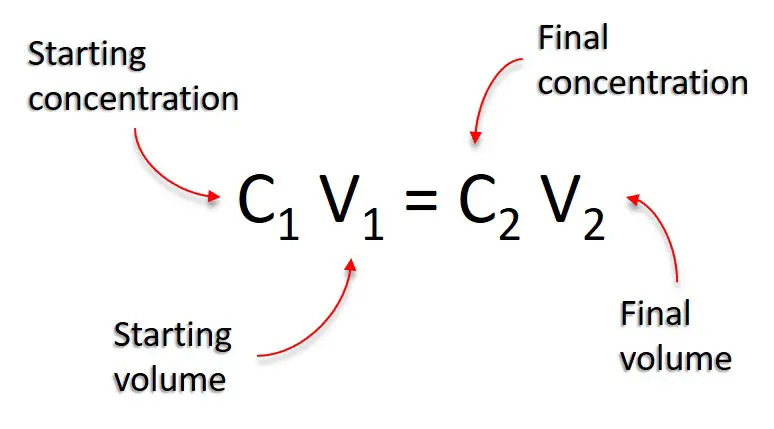
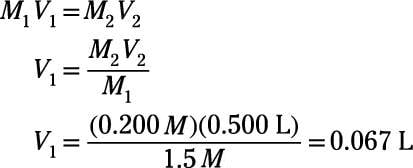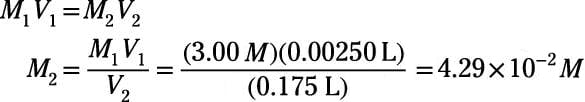What would be the concentration of a solution made by diluting 45.0 mL of 4.2 M KOH to 250 mL? | Socratic

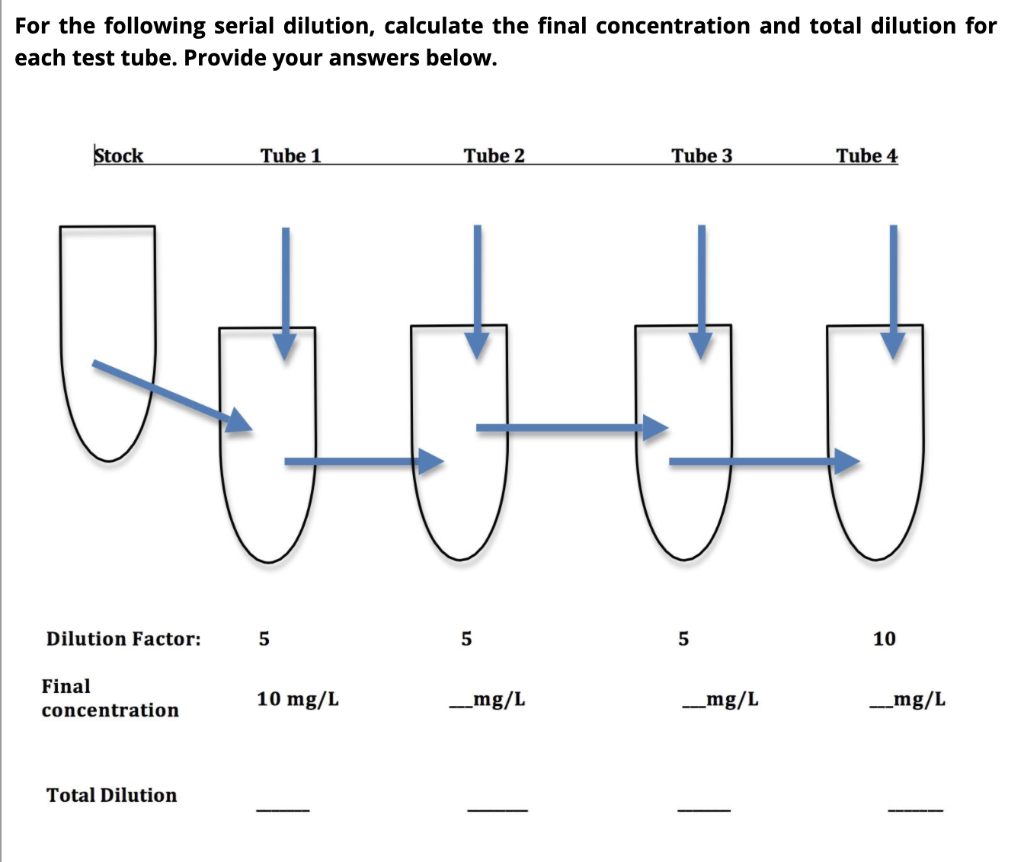
SOLVED: Alternative way to solve dilution problems is to calculate the dilution factor (DF): Dilution factor is defined as total volume of solution per aliquot volume. The aliquot here is concentrated solution,

Dilutions. Solve problems involving the dilution of solutions. Include: dilution of stock solutions, mixing common solutions with different volumes and. - ppt download

Dilution Equation & Examples | How to Calculate Dilution Factors - Video & Lesson Transcript | Study.com

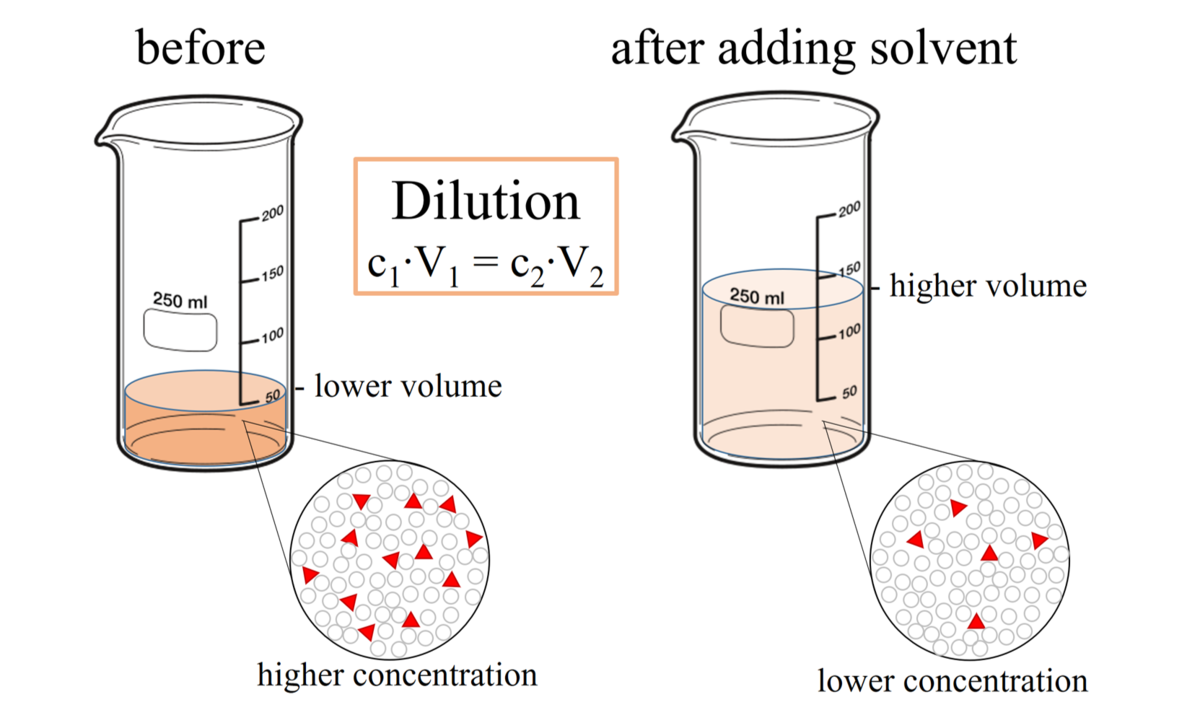
Solution Dilutions. D ilution In a dilution water is added. volume increases. concentration decreases. Copyright © 2009 by Pearson Education, Inc. - ppt download